Author(s): Ms. Manisha Agrahari1, Dr. Raj Kamal Sharma2, Dr. Parvez Alam3 & Mr. Imran Khan Teli4
Author Details :
1. Ms. Manisha Agrahari, Assistant Professor, Department Of Nursing, Radiant Institute Of Nursing And Paramedical Science, Kishanganj, Bihar. manisha_agrahari@rediffmail.com (Correspondence Author)
2. Dr. Raj Kamal Sharma, Associate Professor, Department Of Morphology, Karaganda Medical University, Karaganda, Kazakasthan.
3. Dr. Parvez Alam, Associate Professor, Department Of Pharmacy, Radiant Institute Of Nursing And Paramedical Science, Kishanganj, Bihar.
4. Mr. Imran Khan Teli, Principal, Department Of Nursing, Radiant Institute Of Nursing And Paramedical Science, Kishanganj, Bihar.
Published In: Volume – 3, Issue – 1, Year – 2025 (JAN-JUN)
How to cite this article?
Agrahari M, Sharma RK, Alam P, Teli IK. A descriptive study A Study to Assess the Effectiveness of Lingamudra on Respiratory Discomfort Among College Students in Selected Colleges, Coimbatore.
Abstract:
Children with prosopagnosia, also known as face blindness, struggle to recognize familiar faces, which can negatively impact their social interactions and everyday functioning. Recent advancements in array-based technologies and high-throughput sequencing have made comprehensive genome-wide analysis of ASD loci possible. Whole-exome capture has revealed that known protein-coding sequences make up about 1% of the genome. This overview highlights significant findings from key strategies in ASD genetics. Although there is no prevalence estimate for autism, it is suggested that 2-3% of the general population experiences developmental prosopagnosia, or difficulty with face recognition. Research on developmental prosopagnosia (DP) often uses autism symptoms as exclusion criteria. We examined whether 43 individuals with DP, who had varying autism quotient (AQ) scores, processed faces differently due to autistic traits. Both the high-AQ and low-AQ DP groups showed deficits in face memory and perception; however, the high-AQ DP group also exhibited difficulties in recognizing facial emotions. Meta-analyses and case-control studies indicate a moderate level of impairment in this group, with only a few studies finding no impairment. One possible explanation for these mixed findings is that variability in face-processing abilities may link some prosopagnosic individuals with ASD to those who manage daily situations. Randomly selected community samples, especially smaller ones, may show varying degrees of prosopagnosia, leading to group-level deficits ranging from mild to severe. Additionally, certain genes associated with various psychiatric traits and related neurodevelopmental disorders appear to be risk factors for ASD. Genes linked to ASD include those related to fragile X mental retardation-associated proteins and synaptic proteins like SHANKs, neuroligins, and neurexins. Global regulation of gene controllers and chromatin modification are examples of ASD genes that lack a clear mechanism of specific neurological dysfunction. The combined effect of strong-impact genetic disorders and weak-impact common genetic variants has not yet provided a cohesive understanding of ASD pathophysiology or explained its wide range of clinical manifestations, despite significant and rapid advancements in genetic research. Prosopagnosia was prevalent among nearly one-third of the autistic participants. Their general cognitive abilities, empathy, alexithymia, and symptom severity were not linked to their facial memory skills. Only autistic individuals with prosopagnosia showed a connection between face recognition and mental state recognition from the eye region; this association was independent of participants’ basic face recognition abilities. Importantly, we discovered that those with autism were unaware of their face memory skills.
Keywords: SHANKs, autism spectrum disease, neuroligins, neurexins, prosopagnosia, developmental prosopagnosia, and ASD genes.
Introduction:
The neurodegenerative disease known as a lack of interest, repetitive activities, and difficulties with social communication are characteristics of autism spectrum illness (ASD) (Hyman et al. Citation2020).Data analysis from the Centres for Disease Control and Prevention in the United States, one out of every 36 children has autistic spectrum disorders, up from two years before (Maenner et al. Citation2023). The occurrence of Autism has been more prevalent in the last two decades. according to surveys. However, because to variations in sample size, age, diagnostic criteria, and diagnostic techniques, prevalence survey statistics might vary significantly. Individuals with developing prosopagnosia develop recognition issues in the absence of evident brain damage, as opposed to acquired prosopagnosia, which occurs as an adult after a stroke or a traffic accident. Persons with developing prosopagnosia struggle to recognise faces even when their IQ, memory, and normal low-level vision are all acceptable.
The key research topic in DP is whether the disease impacts aknowledgment (domain-specific) of only faces or a wider range of visual characteristics. Geskin and Behrmann (2018) found in a comprehensive meta-analysis that 80% of DP people had relevant question prosopagnosia, which supports a domain-general theory. Acknowledgment issues were shifted, affecting a wide run of thing categories, counting autos, scenes, buildings, blossoms, glasses, shells, weapons, steeds, devices, and obscure items.
Many patients with prosopagnosia are aware of their problem. Although some people come up with compensatory techniques, including recognising someone by their voice, many patients are upset and ashamed of their disability. Finding the anatomical correlate of prosopagnosia was the main focus of the study for the first hundred years following its identification as a neurological disease. The medial occipito-temporal cortex’s lingual and fusiform gyri were the centre of the phase, however it was not apparent if the involvement was bilateral or right unilateral.
of gene mutation in developmental prosopagnosia with children suffering from autism spectrum disorder. Int J Adv Res Med Nurs Health Sci. 2025;3(3):70-83. Available from: http://www.ijarmnhs.in.
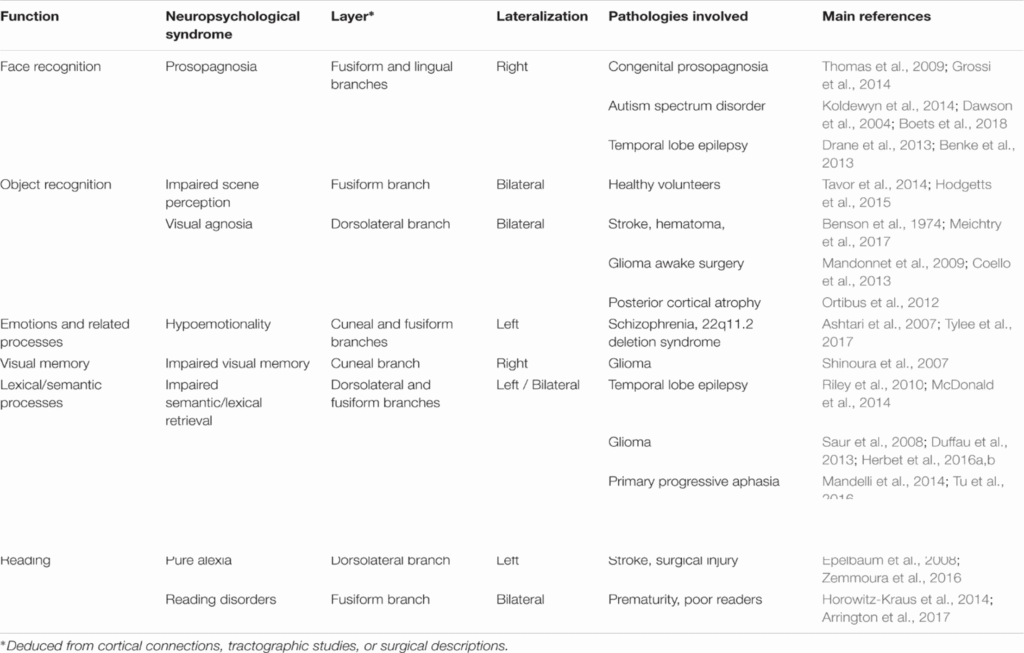
Table 1.
At first, there was disagreement about whether CT imaging required involvement of both hemispheres or a localised lesion in the right hemisphere. However, as its usage in neurological disorders has grown, it appears that a single lesion in the right hemisphere is adequate . De-Renzi and colleagues proposed a classification of prosopagnosia into two types over the next few decades: apperceptive and associative (amnestic). Additionally, Warrington and colleagues demonstrated that facial recognition loss is distinct from loss of face discrimination and might be exclusive to human faces. It is probably polygenic, can run in families, and seems to be impacted by the OXY system. The most reliable method for evaluating faces that are new to the subject is the Cambridge Face Memory Test (CFMT), which offers a clinical cut-off value for prosopagnosia.

Figure 1. Cambrige Face Memory Test
It is generally agreed that objective assessments of face IIR should be a significant part of any diagnostic evaluation, even though there are no specific diagnostic criteria for DP.
Research highlights early warning signs to start early management because there are no molecular indications for diagnosis. An review of the theoretical relevance of early motor development signals is given by Posar and Visconti. According to their suggestion, motor and communication development is interdependent, and their dysfunction is a crucial aspect of autism’s pathophysiology. Motor disturbances in these children can occur due to early motor development delays or the occurrence of aberrant movements. These modifications may serve as warning signs, prompting treatments that aim to target common or proximal brain areas for minimally affecting ASD symptoms through early normalization of motor development. Proprioceptive sensory dysfunction leads to bodily instability in autistic children, as demonstrated by Abdel Ghafar et al.
Current research supports the efficacy of early intervention strategies that utilize principles of behavior analysis and child development, including naturalistic developmental behavioral interventions (NDBI) that focus on other things, normal physiological principles, ecological settings, regular development, sensory regulation, and organised learning environments. The most concerning aspect pertains to the genetics of Disorder of the Autism Spectrum (ASD). Despite the high heritability rate of ASD, which lends credence to the notion that autism has a genetic basis, research repeatedly demonstrates that only a small proportion of adolescents with Autism have a distinct genetic aetiology.
One possible explanation is that the genetic factors involved are more complex than previously thought, leaving us with an incomplete understanding. Moreover, studies show that 69% of twins with ASD and genetic abnormalities have distinct genetic mutations, and most of the genetic changes found are de novo, suggesting that transmitted genetic mutations could not be the main cause of the exceptionally high heritability rate. Further research indicates that both environmental and genetic factors likely combine to produce ASD, with both aspects contributing significantly.
However, research on developmental prosopagnosia (DP) has used a range of tasks, including the face reverse task (Klargaard et al., 2018), the part-whole task (DeGutis et al., 2012), and, to a lesser extent, the combination of face assignment (Avidan et al., 2011; Palermo et al., 2011; see Biotti et al., 2017 for an exception), to show a consistent pattern of deficiencies in holistic processing. According to research, in comparison to developmental prosopagnosia (DP), autism spectrum disorder (ASD) is often associated with more widespread network abnormalities at the brain level (Uddin et al., 2013). The fronto-parietal and posterior superior temporal sulcus, which are essential for deciphering social intentions and behaviours, are less connected in people with autism spectrum disorder (ASD). Temporal parietal junction activity is reduced in fMRI studies, which helps with “theory of mind” while watching socially unpleasant stimuli.
Compared to a control group and a prosopagnosia group, two-thirds of individuals with social developmental disorder (SDD) show difficulties with facial recognition, according to a study by Barton et al. (2004). On the Social Skills Inventory, these participants did not exhibit any recognisable changes, indicating that face processing deficiencies have no bearing on the degree of social-emotional difficulties in the SDD group.
Genetic Exploration
Recent studies have used cutting-edge techniques and technology, including as molecular microarrays and parallel sequencing, to identify a large number of inherited risk factors for ASD, including distinct risk loci spanning genomic regions and individual nucleotides. Due to the limited sample sizes in ASD, many novel loci require more study and more reproducible statistical significance.
Due to their association with other illnesses and the identification of rigorous chromosomal-wide surveys of particular polymorphisms and genome structure, the genetic architecture of ASD was previously unknown. Additionally, despite being mostly unreliable due to numerous false-positive results, candidate gene studies did uncover links with a small number of genes that are still linked to ASD in some research.
The text outlines various types of genetic variations (such as SNPs, CNVs, translocations, inversions, and inndels) and their inheritance or de novo status differ and how frequently they occur in the population (rare vs. common). Here, we provide an overview of the genetic risk for autism derived from transcriptomics, epigenomics, common and rare genetic variation research, and linkage and candidate gene studies.
Unbiased genome-wide evaluation of ASD loci is now possible thanks to the development of array-based technologies and massively parallel sequencing. This leads to whole-exome capture, which looks at recognised protein-coding sequences in the 1% of the genome. The most notable results from key methods in ASD genetics are highlighted in this evaluation.
Analysis Of Potential Gene And Its Linkage
Twin studies conducted over the past 50 years have shown that monozygotic twins had an ASD concordance of 50–90%, but dizygotic twins have a 30% concordance. It is now acknowledged that ASD is a highly heritable disorder, and that the risk increases with the number of afflicted family members . They include common genetic variants, mosaicism, inherited recessive variations, non-coding and regulatory harmful alterations, copy number variants (CNVs), and de novo variants with a single letter (SNVs).
Instead, there is now no known genetic cause for about 70% of the afflicted individuals. There is a link between phenotype and gene dosage, but confirming the relationship is sometimes challenging. De novo sub microscopic structural variation was found to be significantly associated with autism in 2007 using comparative genomic the integration. Since then, other CNVs associated with autism have been found. Trio exome sequencing in clinical settings has demonstrated a noteworthy role in the identification of de novo SNVs associated with autism risk. Given that de novo SNVs connected to autism frequently affect a single gene, this is particularly important when addressing the underlying neurology of these changes. Evolutionary constraints are used in advanced bioinformatics algorithms to identify risk genes with a rate of incorrect identification of not more than or equal to 0.1.
Researchers can find mutations influencing gene functions by projected effect in addition to using PolyPhen-2 constraint score, missense badness, and probability of loss of function (pLI). These methods validate predominance of newly generated loss-of-function mutations affecting highly restricted genes and also identify deleterious missense mutations.
Furthermore, in order to better understand the mechanism of pathogenicity, functional experiments are essential for these validations. Comprehensive and up-to-date information on genes linked to ASD may be found in the SFARI gene database 1003 genes are categorised in the 2020 Q4 database as having a strong candidate, high confidence, and persuasive evidence for their involvement in the development of ASD. The risk genes are biassed in their distribution on each chromosome, according to the gene list. The majority of genes associated with ASD are located on chromosome X, which has been proven to validate the male-to-female ASD ratio of approximately 4 to 1.Whole-genome sequencing (WGS) and transcriptome analysis have revealed that certain molecular processes, such as chromatin modifications, transcription of RNA, demonstrating processes, gene transcription supervision, neuronal interaction, cytoskeletal organisation, and cell cycling, may be dysregulated in ASD risk genes. SNVs have been curated with 889 ASD risk genes from SFARI.
Research on Autism Gene Expression by Tissue Type
The categorisation by sample source will be useful when examining the new gene expression experiments that have been conducted since Voineagu’s study. Adult olfactory stem cells, gastrointestinal tissue, post-mortem brain, peripheral blood, and scalp hair follicles were the five sample sources that were examined. It is crucial to discuss gross pathological and radiological brain abnormalities before starting the post-mortem brain tissue research, since these may offer context for the findings of brain-related expression. A manually compiled records of genes connected to ASD (SFARI Gene) and an integrative medicine tool (Autworks) to gather information on autism future research genes alongside those linked to neuropsychiatric disorders, along with an updated set of predicted gene interactions, were used to assess the biological in nature environment and the possible value of the Root 66 cluster. We manually looked for lists of variations connected to autism proposed genes in order to increase the quantity and dependability of the variant data utilised to confirm the physiological usefulness of overall Rooted 66 gene collection. We looked into how facial memory and the eye region’s capacity to identify psychological states are related because both are compromised in autism and depend on information extraction compared to the identical region, which is crucial for both diagnosis and prognosis.
According to our research, the capacity to determine someone else’s emotional state by looking into their eyes was associated with identity identification, which serves as crucial for interacting with others. Notably, only in autistic subjects who suffered prosopagnosia was facial identification recognition linked to mental state knowledge. Considering that non-autistic people with progressive prosopagnosia do not have difficulties identifying their emotions from the eye area, this association appeared unlikely to result through a decreased proficiency with faces.
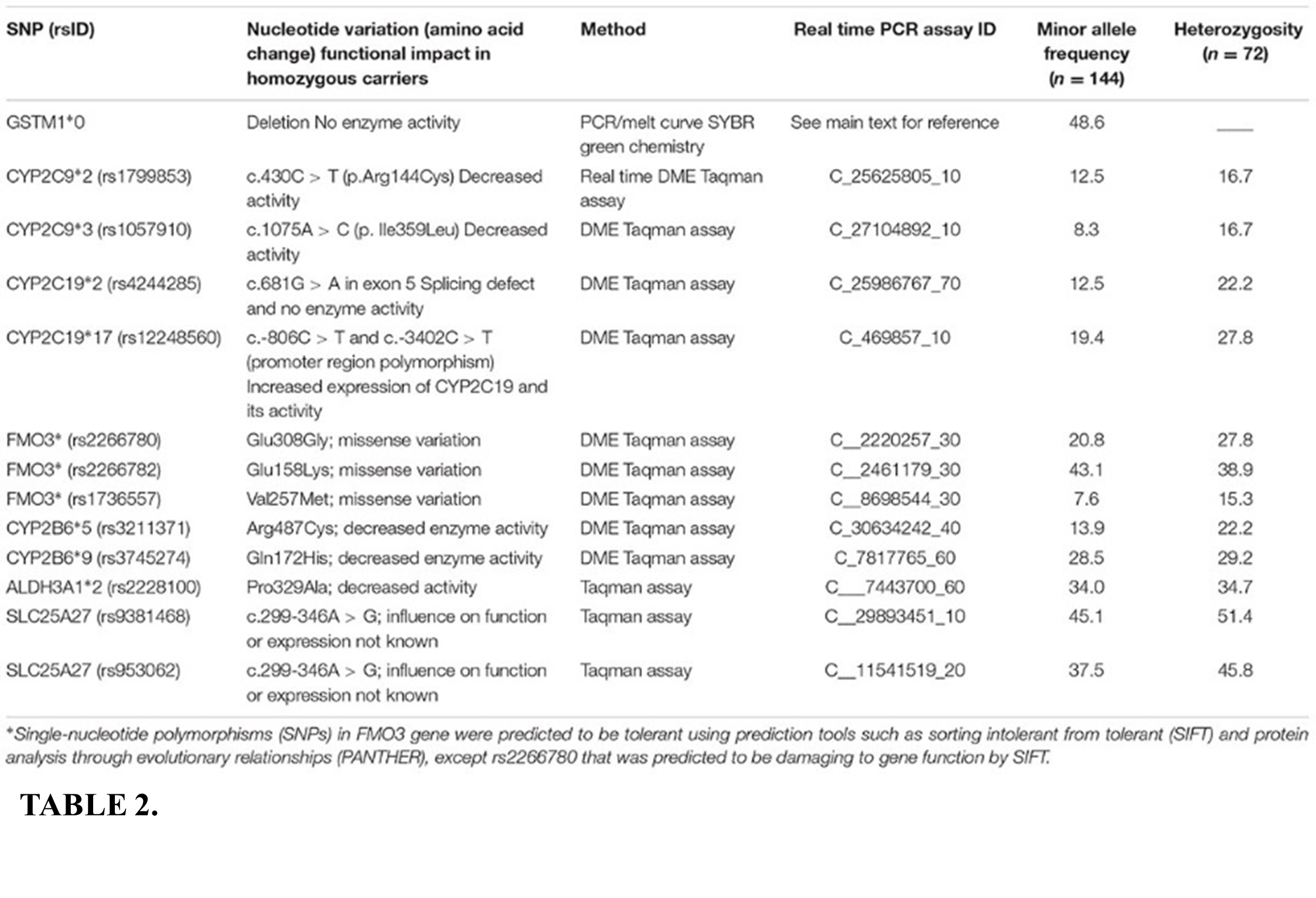
Markers Of Developmental Prosopagnosia In The (In)Visible Brain
In contrast to the acquired variety, DP diagnosis necessitates normal sensory and intellectual skills, as well as face-processing issues that have likely been since birth and are not brought on by any indication of a brain damage. Changes in the structure and connections of the brain have been seen in spite of this. within DP. DPs and HCs may be distinguished using a range of MRI techniques, such as diffusor tensor imaging (DTI), architectural a magnetic resonance imaging and functional connectivity fMRI. The result has made it possible to examine the connections between behavioural and structural data. According to the majority of the evaluated research, the density or volume of the temporal lobes of DPs, namely the pSTS, the meeting has been, and FG, was lower than that of HCs. In the right hemisphere, this type of evidence was more reliable. The core face network had decreased fractional anisotropy and functional connectivity with respect to white matter integrity, particularly next to the r-FFA. These results align with research on the neurological underpinnings of facial processing as well as damage associated with acquired prosopagnosia. The MTG and ITG are linked to the naming and recognition of well-known faces and structures, despite the fact that they are not face-selective (here, semantic memory).
Although their FFA along with OFA grey matter levels do not seem to be reduced, there is mounting evidence of altered white matter inside DPs’ VOTCs. In DPs where communication along the main face circuit is hindered, the r-OFA and r-FFA in particular have emerged as crucial nodes. In particular, the r-FFA primarily exhibits degradation in a long-term functional connectivity during the extended face network, whereas the r-OFA exhibits impairment with regard to short-range as broad operational connectivity within the underlying face network. Subsequent investigations identified many areas in the core- and extended-face networks of DPs that had reduced functional connection to the r-OFA and r-FFA. In terms of DPs, Haeger et al. and Gilaie-Dotan et al. found larger MTGs, although Behrmann et al. observed no structural changes. Two long white matter networks in DPs and their structural integrity was significantly reduced, according to Thomas et al., however these findings have not been validated or improved upon by other research. Lastly, Dinkelackler et al. The study discovered a link between deficiencies in face memory and reduced cortical volume between the left sublingual gyrus and l-DLPFC. Although these two regions are important in both spatial cognition and memory processes, we also note that Dinkelackler’s DPs also exhibited moderate impairments in non-face visual memory. Consequently, this data may indicate a more widespread visual impairment rather than being only attributable to deficiencies in face memory.
Developmental Prosopagnosia: Face-Induced Brain Activity
Research methodologies and findings differ when comparing face-induced activity in the brain in DPs and HCs. Block designs were found to be more common than event-related designs due to the poor temporal accuracy of fMRI blood-oxygenation-level-dependent (BOLD) information the signal-to-noise ratio advantage, as well as the technological constraints of conducting activities in the scanner. Actually, block designs—particularly those that incorporate passive staring and one-back tasks that compare faces to non-face stimuli—provide the majority of the fMRI evidence for DP. Although the former is a perceiving task and the one below is a sequential feature-discrimination assessment with little memory load, both were utilised to examine the neurological underpinnings of face processing in DPs.
The relationship between recognition of facial features and social skills has been the subject of several research. In research conducted by Barton et al. (2004), In order to evaluate any face recognition defects, the investigation compared the face recognition abilities of 12 persons with prosopagnosia and 24 adults with social developmental conditions (SDD) to those of usually developing people. According to the results, compared to the control category (mean = 2.77) and the prosopagnosic group (mean = 0.37), around With d’ scores that varied from 0.75 to 2.25, included a standard deviation of 1.59 on a Prominent Faces Test, two-thirds of the SDD persons struggled with facial recognition.
Interestingly, there was no significant difference in the Social Competency Inventory ratings of SDD participants with face identification difficulties compared to the remaining participants of the SDD cohort (Riggio, 1992).This suggests that the severity of social-emotional difficulties within the SDD group was not influenced by the degree of perception of faces deficits. This suggests that although face processing impairments may often coexist with social developmental abnormalities, they are neither a certain consequence of these disorders nor a function of the severity of the condition.
As of right now, the best model for comprehending how face-related information moves through face-selective regions depends on whether faces are present or not. This affects the feed-forward efficient communication from both the main and the secondary visual nerve endings to the core face network. In face observation networks, developing prosopagnosics (DPs) showed noticeably less connectivity than healthy controls, indicating that these connections could be essential for precise facial recognition. Together, these findings provide valuable information on the neurological processes underlying face processing in DPs and highlight the critical role of the fusiform organism face area (FFA) and its communication with other brain areas in face recognition. The analyzed evidence indicates that individuals the facial representations of people with DP are unusual and fundamentally different from the faces of healthy counterparts (HCs).
By interfering with the grey and white matter associated with the fusiform face region (FFA), parts-based methods can successfully detect unique facial features in DPs. Furthermore, as compared to HCs, DPs with adequate face perception skills exhibit different activation patterns, indicating that certain DPs may acquire compensating mechanisms. To sum up, DPs have anomalies in neurological face representation in the upper right occipital appearance area (r-OFA) and the FFA, indicating difficulties with both feature-based and holistic face processing. The presence of both shared and distinct neural correlates for face memory as well as perception might help explain the various traits of DPs and the corresponding scientific discoveries. Still, there are a lot of uncertainties that remain.
Taking Development Into Account While Exploring Cognitive Mechanism
The elevated co-occurrence rate of developmental prosopagnosia (DP) and object agnosia, along with the diverse challenges faced by individuals with DP, are fundamental characteristics of developmental disorders in general, where the co-occurrence of various difficulties is more common than rare . Instead than restricting participant selection, evaluations, and data collection to certain diagnostic categories, developmental scientists are increasingly attempting to explore the underlying causes of neurodevelopmental variations (Astle & Fletcher-Watson, 2020).
Despite the effective use of transdiagnostic techniques to address neurodiversity (Astle et al., 2019; Mareva & Holmes, 2019), little attention has been paid to the components of developmental prosopagnosia (DP) and face recognition skills. Even though most people believe that DP is diverse, recruitment, study design, and assessment of research studies have not taken this knowledge into account. Considering this variation while developing the inclusion along with exclusion criteria for choosing the samples is an important first step.
Embracing a transdiagnostic strategy in recruitment would enhance sample diversity, facilitating a more comprehensive understanding of individuals who have difficulty recognizing faces, rather than solely focusing on specific visual recognition impairments. In this context, it would be beneficial to incorporate scores from face recognition assessments along with developmentally pertinent measures that account for co-existing challenges. Instead of applying rigid exclusion criteria, we propose that recruitment should begin with individuals who self-identify as having face recognition difficulties. Future initiatives should concentrate on the differences in performance in psychology of development by looking at the sample’s co-occurring challenge patterns. A broad range of evaluations including different visual categories and any neurodevelopmental problems should be part of this. Neurodevelopmental profiles of challenges can be exposed through data-driven clustering investigations, which identify emergent groupings based on characteristics across several factors (Astle et al., 2019).In the first place, this can assist in measuring the frequency of co-occurrence of obstacles and exploring the connections between them outside of conventional diagnostic classifications.
Conclusion
The condition is thought to have a genetic foundation and is regarded as heterogeneous, with several underlying causes. The exact onset of DP is still uncertain, and the prognosis for individuals on the spectrum is affected by factors such as age, level of impairment, and the impact of various treatments. Treatments linked to DP are strongly associated with face recognition abilities, but autism-related traits do not predict face recognition skills when considering prosopagnosia traits. This highlights the necessity for further research on visual perception in ASD, and developmental prosopagnosia (DP) will be explored. Reevaluating the developmental origins of differences in face recognition abilities is essential to deepen our understanding of DP, rather than focusing solely on outcomes. The presence and variety of challenges linked to DP are fundamental aspects of developmental disorders, where the coexistence of difficulties is common. Prosopagnosia may be congenital or develop later due to various neurological conditions, including neurodegenerative diseases, focal brain injuries, non-degenerative disorders like epilepsy and migraines, and functional disorders. Difficulties in recognizing interpersonal information (IIR) may contribute to the genetic predisposition of autism, similar to a multivariate correlated liability model that includes various genes, endophenotypes, and environmental factors. Studying face IIR could lead to the identification of new genetically relevant subgroups of autism, neurobiological mechanisms, and neural systems. Since both facial recognition and the ability to discern mental states from the eye region are impaired in autism and rely on data from the same source, the connection between the two was examined. As this relationship was only found in autistic individuals with DP, it is unlikely to be due to a lack of facial familiarity.
References